EHA congress 2025
From 12th to 15th June, ALAN attended the European Hematology Association (EHA) 2025 congress, held in Milan, Italy. Below is a summary of the activities in which ALAN took part, in addition to meeting with our members, sponsors and with the medical community.
EHA congress 2025, by Charles, ALAN Steering Committee member and EHA AYA Taskforce
My second EHA Congress 2025 offered a range of insights into the latest developments in haematology, many of which placed the patient front and centre of the agenda: a fantastic step forward for the haematology community.
My focus for Congress this year was on preparing a session on communicating with adolescent and young adult (AYA) patients, particularly in terms of sexual and reproductive health. Fellow AYA advocate Yunus Borowczak (CML Advocates Network) delivered an inspiring and powerful presentation on the dos and don’ts of communicating with AYA patients, covering issues such as transitioning from paediatric to adult care, the best way for clinicians to engage with young patients in face to face consultations, and the best methods of information sharing during and beyond treatment.
We heard from Dr Adam Duvall of the University of Chicago on an online tool for use by multidisciplinary teams to advise AYA patients giving advice and support on sexual and reproductive health following a haematology diagnosis. Both Yunus and I then contributed to a panel discussion which covered the psychological effects of disease and treatment, particularly in terms of relationship forming and maintaining (e.g. when is the best time to tell a new partner you have or have had leukaemia) and the disparities in sexual health support within and across healthcare systems.
Other insights from Congress included sessions on:
- Patient reported outcomes (PROs) in clinical trials and their importance in determining care pathways, and how AI and Large Language Models are making qualitative data far easier to categorise and quantify
- A specialised working group (SWG) covering advances in combination therapy and finding the right drugs for older AML patients
- Measurable residual disease as a surrogate endpoint in clinical trials, and the disparity in its use for different haematological malignancies (very well presented by ALAN’s very own Anne-Pierre Pickaert!)
- And – in a slightly left field move – a session on the long-term consequences of space travel on haematopoeisis
In between the sessions, I reconnected with fellow ALAN members – a particular highlight being a poster presentation on ALAN’s recent study on acute leukaemia caregivers’ Quality of Life. That’s not to mention the Community Leadership Dinner focused on the specific unmet needs of AYA patients and a get together with this year’s cohort of European Patient Advocacy Institute (EPAI) advocates!
Overall, Congress was as fulfilling and insightful as ever, with many signs of progress in putting patient voices at the heart of haematology. While these sessions demonstrating progress were hopeful, I always find that the most rewarding aspect of these events is connecting with others who have been through similar experiences to my own and seeing us all do our best to represent the interests and values of patients touched by haematology, be they patients or caregivers.
Onwards to Stockholm!



What really matters to acute leukemia patients when they survive?
Patient/carer-guided quality of life and preferences research is needed to judge patient reality and needs.
ALAN is a pioneer in community-run patient preference studies. We ran Discrete Choice Experiments, Best-Worst-Scaling, online bulletin boards and interviews with more than 800 patients and carers.
Interestingly, survival at any price is not everyone’s priority: subgroups of acute leukemia patients are willing to trade treatment response against quality of life.
Also, in terms of quality of life attributes, pain, cognitive impairment and tiredness scored highly, with the latter two not measured by EQ-5D.




Assessment of demographics, information provision and involvement in decisions on QoL in patients with leukemia
In this primary analysis, we examined how:
– demographics,
– information provision, and
– involvement in treatment decisions
are associated with QoL impact in patients with leukemia.
The data show that younger leukemia patients and people with acute leukemia types experience worse quality of life and high psychosocial burden. Additionnally, clear communication and shared decision-making significantly improves many aspects of daily living.
Our recommendations for physicians and their daily practise is therefore:
– to have a clear & compassionate communication
– to actively involve their patients in decisions, and
– to be more mindful with acute leukemia and younger patients who require additional support.
The poster presented also show that increased patient engagement in treatment decisions, clarity of information on treatment side effects and transparent communication may serve as protective factors against the psychosocial burden of leukaemia, suggesting the important role of patient support groups.



Representing Patient Perspectives on MRD at the EHA-EMA Joint Symposium
On Friday, June 13, 2025, Anne-Pierre Pickaert represented the Acute Leukemia Advocates Network (ALAN) as a panelist at the EHA-EMA Joint Symposium during the European Hematology Association (EHA) Congress in Madrid. The session, titled “Minimal Residual Disease (MRD) in Clinical and Regulatory Decision Making,” brought the perspective of a clinician, a regulator, a researcher, and a patient advocate to explore how MRD is shaping decisions across the care, research, and policy landscapes in hematologic malignancies.
A Thoughtful, Multi-Perspective Dialogue
The session, chaired by Dr. Pierre Démolis of the European Medicines Agency, also featured:
- Prof. Dr. med. Nicola Gökbuget (Frankfurt University Cancer Center), who shared a clinical perspective on MRD in acute lymphoblastic leukemia
- Anna Smit (Erasmus MC), who presented early data from a survey on MRD use in multiple myeloma
- And Anne-Pierre Pickaert, who contributed insights grounded in her advocacy work with two French patient organisations: Association Laurette Fugain, a proud ALAN member, and EGMOS (Entraide aux Greffés de Moelle Osseuse), which supports bone marrow transplant recipients.
This mix of perspectives created space for genuine dialogue on the opportunities and challenges of integrating MRD into both practice and policy. A recurring theme throughout the discussion was the importance of including patient voices early — not only to shape clinical trial design and regulatory frameworks but to ensure that science remains grounded in the lived experience of those it seeks to serve.
Bringing Forward the Patient Voice
In her remarks, Anne-Pierre shared how individual acute leukemia patients see MRD as a meaningful predictor — not only of remission or relapse, but also of eligibility for bone marrow transplant. However, she pointed out that MRD awareness remains uneven across the blood cancer community. While MRD is more routinely used in diseases such as AML, ALL, CML, and multiple myeloma to guide treatment decisions, it is not yet integrated into standard care for lymphoma and CLL. In these areas, patient advocates are only beginning to engage with MRD as a research endpoint to assess short-term treatment effects.
This inconsistency poses communication challenges for patient organisations that support multiple blood cancer communities. They must adapt their messaging to reflect the varying clinical relevance — and limitations — of MRD for different diagnoses.
Reframing MRD in the Regulatory Space
Anne-Pierre also addressed how current reliance on overall survival (OS) as the primary endpoint for drug approval can delay patient access to innovative therapies — particularly in fast-progressing diseases where waiting for OS data is not practical. MRD could offer a meaningful early signal of treatment efficacy if consistent thresholds and assessment guidelines are adopted globally and OS continues to be collected as a co-primary endpoint.
She welcomed Pierre Demolis’s perspective that instead of pursuing MRD as a validated surrogate endpoint — a path that is both methodologically complex and time-consuming — it should be framed as a “response assessment.” This framing reflects MRD’s ability to demonstrate a drug’s biological activity and potential benefit more quickly. Shifting terminology in this way helps regulators and stakeholders stay focused on clinical relevance and timely access — rather than getting mired in technical validation processes.
Extending the Conversation Beyond the Congress
The discussion was later captured in a Medscape feature article by journalist Cristina Ferrario, titled “MRD: A Shared Compass for Patients, Clinicians, and Regulators.” he article reflects the panel’s key messages — including the diverse perspectives on MRD’s use, the regulatory challenges surrounding its validation, and the importance of incorporating patient experience into the evolving conversation on measurable disease response.
Report on EHA-Patient Joint Symposium – by Sophie Wintrich, ALAN Steering Committee member
The European Haematology Association (EHA) Annual Congress stands as a cornerstone event in the global haematology calendar, bringing together clinicians, researchers, industry leaders, policy makers, and—critically—patients and patient advocates. The EHA-Patient Symposium, and other sessions held during EHA2025, continued its tradition of promoting meaningful dialogue and shared learning between all stakeholders. This year’s series of sessions were designed not only to inform, but to empower and inspire, facilitating a collaborative approach to advancing care, research, and awareness in haematology.
EHA Patient Joint Symposium
The EHA-Patient Symposium at EHA2025 was structured around the theme of “Collaborative Innovation: Patients at the Heart of Haematology.” The sessions were carefully curated to reflect the evolving landscape of haematological disorders, the unique challenges faced by patients, and the opportunities for partnership in research, clinical care, and advocacy.
Evolving Endpoint Strategies: Navigating New Therapies and Regulatory Acceptance
Developing standardized and globally accepted endpoints, including quality of life (QoL) as a co-primary endpoint, is essential for assessing the value of new drugs in both malignant and non-malignant hematology. This session delves into the perspective of clinicians, patients, industry, and regulators with regard to defining and standardizing endpoints for treatment evaluation. Panelists will discuss the limitations associated with traditional endpoints and the importance of Real-World Evidence for complimenting clinical trial data and will share their views on how to incorporate patient-valued outcomes and adopt a holistic approach.
The session featured speaker presentations, followed by an interactive discussion with the audience.
Panel:
- Jenica Leah, European Sickle Cell Federation (Chair)
- Prof. Lorenzo Brunetti, Università Politecnica delle Marche (Chair)
- Dr. Francesco Pignatti, European Medicines Agency (EMA)
- James Ryan, European Federation of Pharmaceuticals Industries and Associations (EFPIA)
- Dr. Beate Wieseler, Institute for Quality and Efficiency in Health Care (IQWiG)
All speakers were in agreement that involvement of patients in the design of trials would bring additional benefits and clarity.
Patient Advocate Jenica Leah made a very strong impact with her personal experience of living with Sickle Cell – and her recommendations for better comprehensive care, that does not overlook the many QOL issues.

Key messages:
- End points should include clinical data AND PROs
- Outcome data should by looked at by age groups
- RWE needs tracking post-trial
- Trials ought to be designed with patients, not just for them, to ensure they address what patients want
Prof Lorenzo Brunetti specified that surrogate endpoints have limitations and overlook the patients.
As an example – an AML study showed benefit in EFS, no improvement in OS AND significant side-effects – hence no benefit to patients.
On the other hand, the MAYA study in MM included patient data in addition to surrogate end points – and was a success.
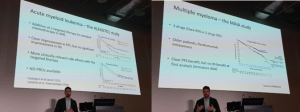
Key message: Surrogate endpoints must be coupled with PROs.
Francesco Pignatti did question the work done by regulators – stating that the patients with their knowledge of the disease should point out the most appropriate end point.
In addition, the acceptance of risk by patients should be used to calculate the risk/benefit.
He suggested we look at overall time patients spend in health, and that patient preference studies may soon inform decisions.

Key message: we must focus on experience
James Ryan, representing EFPIA thought evidence must address diverse needs
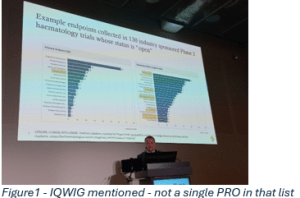

Beate Wieseler – from IQWIG stressed that criteria coming from both patients and healthcare system perspectives need to be used to measure effectiveness and safety, as well as added benefits.
Made a powerful point that ‘We know how to do it – it is not rocket science, but still not happening. And why? Because programmes are still focused on needs of regulators.
Instead, study programmes need to meet the needs of all stakeholders. Surrogates are a mean of accelerating approvals, and need to be thought of from the start, with PROs.
She also advised that endpoints data must also be collected in real life – along the life cycle of the drug.

In the discussion – a few very interesting points emerged – again all pointing to giving way more importance to patient preferences.
That clinicians ought to question the validity of end-points – ideally with patients (something that has been suggested for EHA 2026
There were excellent audience questions
- From advocate A-P Pickaert: Why are QOL endpoints just exploratory? EFPIA explained that quality of the data is not guaranteed – to which IQWIG just stated that it is not more difficult – we just have to do it.
- Dr Ben Kennedy: When will we see a mandatory PPI? IQWIG answered that it is not yet in the pharma legislation…
- Advocate Natacha Bolanos: Co-Primary endpoints? – which Foggi answered by stating that studies should be designed to identify what’s the most important to patients.
Advancing Quality of Life as a Measurable Outcome in Clinical Trials
Quality of life is crucial (QoL) for patients and their carers, as it directly impacts their daily well-being and overall satisfaction with treatment. Yet, over the years, little progress has been made in terms of standardization of QoL parameters. This session aimed to integrate patient-reported outcome measures (PROMs) and patient-reported experience measures (PREMs) into clinical trials, providing a structured approach to enhancing QoL as a measurable outcome.
Panel:
- Loris Brunetta, Thalassaemia International Federation (Chair)
- Dr. Sophie Park, Centre Hospitalier Universitaire de Grenoble
- Dr. Ahu Alanya, European Organisation for Research and Treatment of Cancer (EORTC)
- Mairéad Ní Chonghaile, Haematology Nurses & Healthcare Professionals Group (HNCHP)
- Dr. Paolo Foggi, Italian Medicines Agency (AIFA)
This was a very high interest session – with a very packed room – and chaired by the Italian patient advocate Loris Brunetta.
Essentially, all speakers were in full agreement that patients and advocates absolutely must be engaged in the design and choice of PRO tool for studies. Different tools were discussed that can create engagement and foster discussions between patients and researchers, leading to a hermeneutic approach.
In his introduction as Chair, Patient Advocate Loris Brunetta questioned the current choices of PRO tools. Do patient really understand all parameters, are they matching he research intentions, is a slight improvement in mental health really relevant to them?

In the context of clinical trials and patient engagement, a hermeneutic approach involves creating a dialogue between patients and researchers to interpret and understand patients’ experiences and perspectives. This method emphasizes the importance of context, the co-construction of meaning, and the iterative process of interpretation. By fostering open communication and mutual understanding, a hermeneutic approach aims to ensure that the insights and feedback from patients are accurately captured and integrated into the research process. This can lead to more meaningful and relevant outcomes, ultimately improving the quality of life for patients.
A question was posed regarding whether regulators would accept this approach, highlighting the need for regulatory bodies to recognize and validate the importance of QoL measures in clinical trials.
Patient Input in Surveys: It was noted that almost all surveys about QoL are conducted without patient input, leading to problems of perceptions. This underscores the necessity of involving patients in the design and implementation of QoL surveys to ensure their accuracy and relevance.
Dr. Sophie Park, a haematologist in Grenoble, discussed whether current QOL tools adequately capture fatigue in MDS patients. (EORTC-QLQ-C30, FACT-G, FACT-An/F, CTCAE, PRO-CTCAE)
She is aiming to use AI to improve fatigue measurement. In MDS, she found that EQ-5D and fatigue VAS scores do not correlate linearly with HB levels, while QUALMS-P and QUALMS-BF (38 items) show improvement as HB. The current aim is to integrate the QOL measurement into AI chatbots, while preserving clinical validity in such conversational formats.
She checked for compatibility of QOL questionnaires for fatigue and chatbot potential.
In conclusion – there is no perfect tool – as you need to look at exact goals and context. PRO CTCAE remains a standard – and good for chatbot use. However, again – it lacks patient perspective.
Chatbots could provide united views between patients and clinicians, but it will be critical to have validation, ethics approval, patient consent and data protection. Thorough real-life testing will be much needed.

Ahu Alanya from EORTC (Brussels) questioned the rationale for using specific PROs and whether PROs tell the full story. For instance the objective can be HR QOL benefit OR Tolerability of a treatment.

In a real-life example, she reported that a higher rate of AE did not result in worse outcomes.
However – 40% of patients discontinued treatment due to reported QOL issues.
So the solution (and general recommendation from SISAQOL9IMI) )is to include additional analysis to look at patients who discontinued treatment.
PRO Graphs should provide information on sample sizes, intercurrent events and missing data.

Mairéad Ní Chonghaile, Haematology Nurses & Healthcare Professionals Group (HNCHP) highlighted that QOL is crucial – in light of competing treatment options. 50% of patient would not choose additional treatment for any gain of PFS. Such tools improve decision making.
QOL tools need to be specific to disease and the population – A tailored approach – as 1 size does not fit all. We also need to be conscious of the data collection burden on patients, caregivers – as well as staff burdens on time and effort and to remember that nurses on the coal face have no extra time.
We need to balance the usefulness vs a potential tendency for a tick-box exercise (just because it is required by regulators).
Keep it meaningful – and engage with patient advocates around the choice of such tools.

Dr. Paolo Foggi, Italian Medicines Agency (AIFA) had an opening statement which was crystal clear: Patients should be at the centre of clinical evidence generation’. Patient engagement is essential, at CHMP level too
He pushed for use of wearables for continuous monitoring – but stated that regulators don’t have strict recommendations on what QOL tools to use. His advice was to ‘Think of the tools to use well in advance’
He also advised to have plans for tackling missing data, and reminded the audience to have an early engagement with regulators to discuss collection of PROs
Interestingly – he specified that you can use a PRO even if it is not ‘qualified’, as long as it is justified.
Lastly, he pointed out that regulators too, must be transparent in their decisions and labels.

Questions from the advocate audience stated that 80% of trials include PRO data – however only a portion of that data is included in the label.
And French advocate Anne-Pierre Pickaert asked why there were such poor options for patient engagement in HTA in Italy.
In the subsequent discussion, the panel stressed the importance of taking action early – as it is not possible to fix things later (P.Foggi) and Alanya spoke of ‘Common sense Oncology’.
PROs as secondary endpoints were mentioned – for randomised trials
Mairead stated that chatbots and free text analysis will really gain in importance – and that nurses already use thematic analysis to look at such data. She stressed that a difference of 1 point of a fatigue scale can make a major difference in terms of life functions
Also mentioned – that missing information is mostly due to admin errors, not lack of patient participation.
Loris Brunetta concluded the session by underlining that we need a new way to capture data – and especially in the low literacy patients.
Modernizing Tolerability Assessment and Safety Reporting
Modernizing how adverse events and tolerability are reported and assessed is very important for patient safety. It is also crucial for improving the efficiency and quality of clinical trials and for regulatory decision making. This multi-stakeholder session will explore whether the dual goals of advancing tolerability assessments and simplifying safety reporting can be achieved simultaneously. The discussion will be led by investigators with key perspectives from regulators, sponsors, and patients.
After the speakers’ presentations, the session will feature a panel discussion, taking questions from the audience to foster an interactive and insightful dialogue.
Panel:
- Prof. Paul Bröckelmann (Chair), Lancet Haematology Commission on Adverse Events in Haematologic Malignancies
- Prof. Martin Dreyling, Coalition for Reducing Bureaucracy in Clinical Trials
- Dr. Emil Cochino, European Medicines Agency (EMA)
- Dr. Elisa Cerri, AbbVie
- Prof. Tarec El-Galaly, Aarhus University Hospital / ACT EU Multi-Stakeholder Platform Advisory Group
- Ananda Plate, European Patient Advocacy Institute (EPAI)

Advocate Ananda Plate made the strongest impact in that session – explaining that tolerability and QOL are co-related, that acceptability of issues changes depending on a patient’s situation, that duration and recurrence of side-effects matter.
She stated that low-grade chronic side-effects can be worse than severe acute ones – citing examples of diarrhoea.
A most important point was that none of her side-effects and long-term toxicities are captured and reported – as there are zero incentives to continue reporting on toxicities after a trial. And this despite the fact that tools exist to collect data.
And that patients will and want to report them, if the side-effects bother them. Basically, the free advice from patient reports is totally disregarded.
EHA Symposium on Patient Communication: Trust Through Dialogue
This dedicated session delved into the crucial topic of communication between clinicians and patients, especially during time-pressured conversations about clinical trials. Entitled Trust Through Dialogue: Balancing Empathy, Clarity, and Autonomy in Time-Pressured Conversations About Clinical Trials, the session featured expert speakers and patient advocates discussing strategies to foster trust and understanding. Practical guidance was provided on how to communicate complex information empathetically yet clearly, respecting patient autonomy while ensuring informed consent. Role-playing exercises and real-world case studies illustrated the challenges of balancing the urgency of clinical trial enrolment with the need for patient-centred communication. Attendees came away with actionable tools for improving dialogue, which is critical for both patient satisfaction and successful trial participation.

Patient Perspectives and Impact
Across all sessions, the voices of patients and caregivers were spotlighted not simply as beneficiaries, but as active agents of change. Testimonies highlighted the emotional and practical realities of living with haematological disorders, the need for clear and compassionate communication, and the power of advocacy in driving innovation. Participants expressed appreciation for the opportunity to network and learn from one another, and many commented on the sense of solidarity fostered by the Symposium.
Conclusion and Future Directions
The EHA-Patient Symposium sessions at EHA2025 reaffirmed the centrality of patient engagement to the future of haematology. As the field continues to evolve, the commitment to partnership, equity, and innovation will be crucial in delivering better outcomes for all. Feedback from attendees will inform the planning of future events, ensuring that the Symposium remains responsive to the changing needs of the haematology community.
By fostering ongoing dialogue, supporting collaborative research, and prioritising the lived experience of patients, the EHA-Patient Symposium at EHA2025 stands as a testament to what can be achieved when all stakeholders come together with a shared vision of progress and hope.
